Vesicle-Cloaked Virus Clusters
|
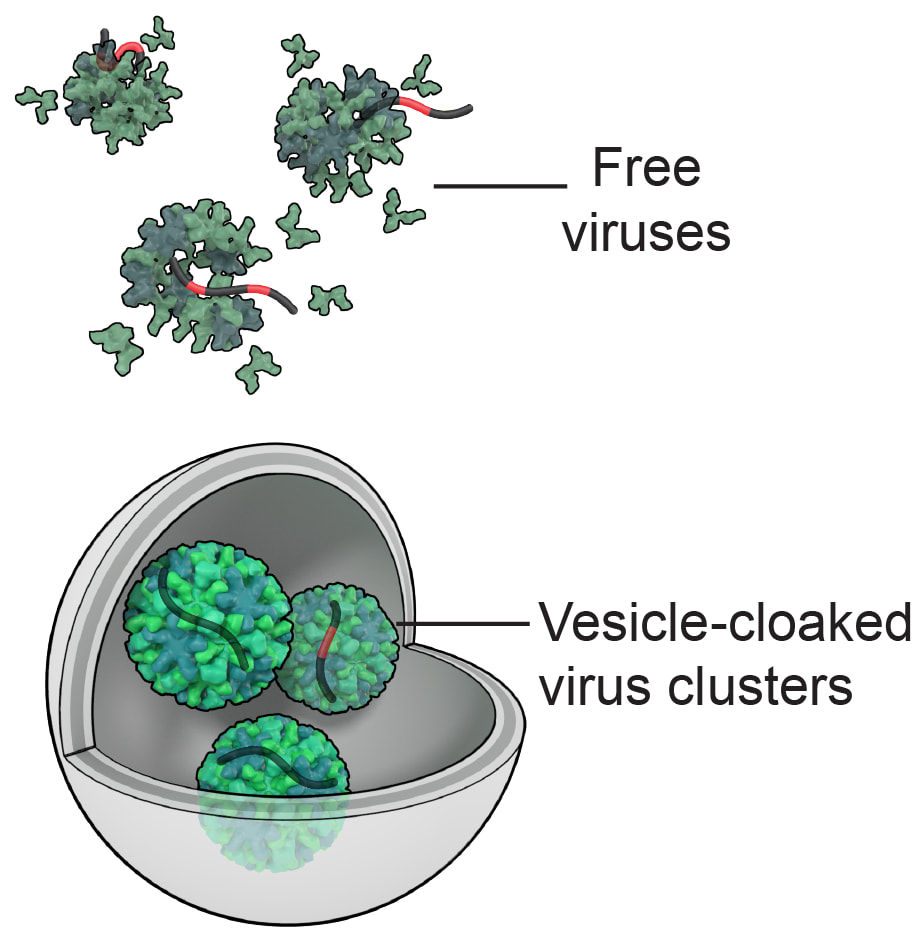
Rotavirus and norovirus, along with many other enteric viruses, can lead to serious gastroenteritis in humans and pose great public health concerns. Water, food, and contact surfaces are always considered as important environmental media that facilitate fecal-to-oral transmission of these viruses. It was widely accepted that viruses were exclusively present in the form of individual particles in transmission and infection, and non-enveloped viruses were released from host cells by cell lysis. However, recent discoveries have challenged this paradigm and revealed that clusters of viruses, including but not limited to norovirus, rotavirus, enteroviruses such as poliovirus, rhinovirus, and the hepatitis A and E viruses, can be packaged and released from their host cells non-lytically in extracellular vesicles. Vesicle-cloaked virus clusters (here referred to as viral vesicles) are phospholipid-bilayer encapsulated fluid sacs that comprise multiple virions or multiple copies of viral genomes, and they can be derived from a variety of cellular origins including autophagosomes, multivesicular bodies, and direct budding from the cell plasma membrane. Extracellular vesicle sizes can range from 100 nm to microns, and different viruses exploit specific-size vesicles derived from specific cellular organelles.
|
Inactivation of free viruses and
vesicle-cloaked virus clusters |
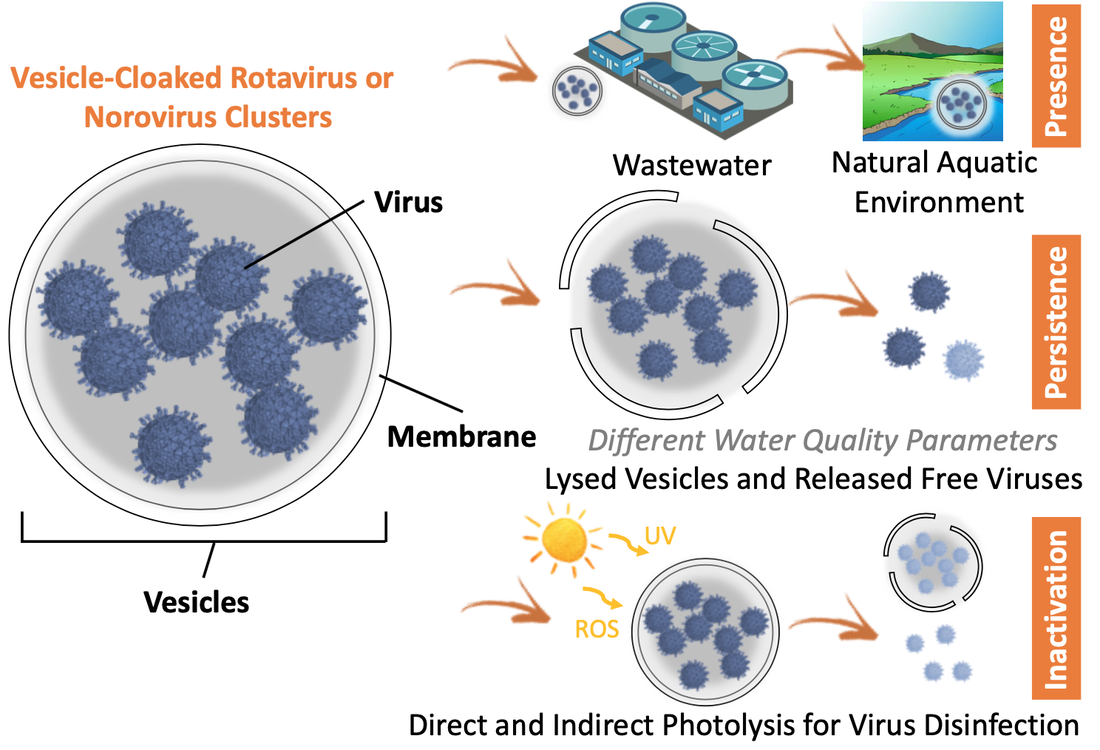
Our previous studies have found that rotavirus and norovirus vesicles are widely present in stool that can contaminate water, they are environmentally persistent in wastewater and surface water, and they are more infectious and are more resistant to disinfection than the counterpart of free viral particles. The discovery of the emerging rotavirus or norovirus vesicles could greatly challenge our previous understanding of the fate, transport, transmission, attenuation, and health risks of viruses in environment. We may face the risk of underestimating the adverse health impacts of rotavirus and norovirus by ignoring the contribution of their vesicles, since almost all previous studies were exclusively conducted by using free viruses.
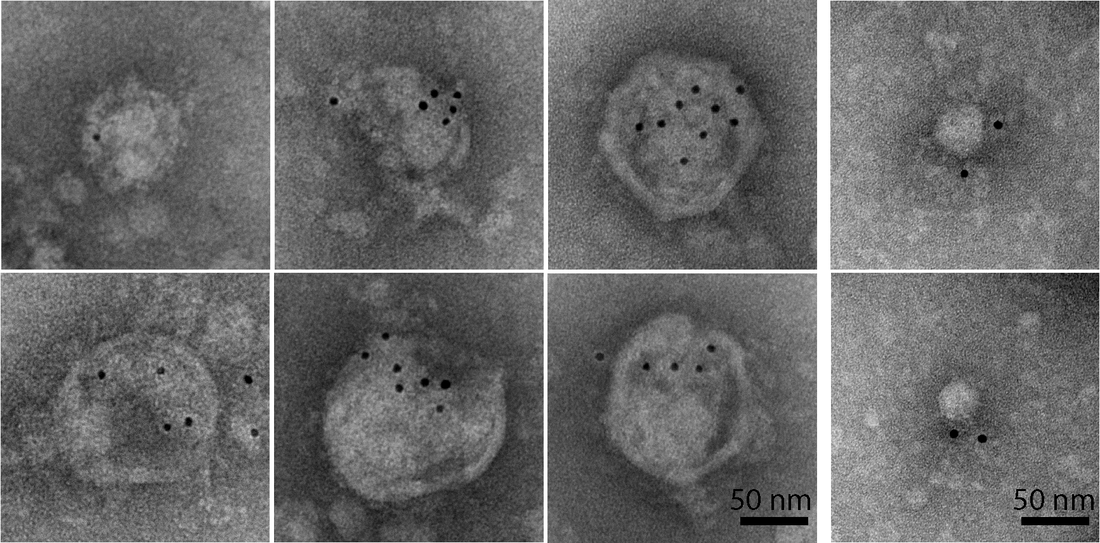
Immuno-electron microscopy of vesicle-cloaked murine norovirus clusters
We are collaborating with Prof. Yun Shen (University of California, Riverside) and Dr. Nihal Altan-Bonnet (NIH) to comprehensively evaluate the fate, transmission, and inactivation of rotavirus and norovirus vesicles in surface water, wastewater, reused water, and soil and on crops and contact surfaces.
Ongoing Projects
Presence, Persistence, and Inactivation of Vesicle-Cloaked Rotavirus or Norovirus Clusters in Water (Funded by NSF-Environmental Engineering Program, 2020-2023)
|
We aim to first understand the presence and persistence of rotavirus and norovirus vesicles in wastewater and surface water. Key water quality parameters that determine the stability of the viral vesicles will be identified. Solar disinfection is an important process that not only inactivates viruses in the natural aquatic environment but also treats contaminated water for reuse. Therefore, we also aim to understand how the viral vesicles are inactivated by sunlight through direct and indirect photolysis.
|
Vesicle-Cloaked Virus Clusters as Emerging Pathogens: Will They Challenge Current Disinfection Paradigm? (Funded by NIH R21 Program, 2021-2023)
|
Disinfectants like detergents, ethanol, bleach, and ultraviolet light (UV) are widely used for household sanitation and contact surface cleaning. However, the performance of these conventional disinfectants for inactivating rotavirus and norovirus vesicles is unknown. Our previous study has found that vesicle-cloaked murine norovirus clusters were more resistant to UV254 disinfection than free murine noroviruses, especially at a low viral load that is more environmentally relevant. In this study, we aim to understand the performance and mechanism of conventional household disinfectants for inactivating rotavirus and norovirus vesicles. Moreover, we will advance disinfection methods and evaluate the performance of emerging disinfectants for inactivating these viral vesicles.
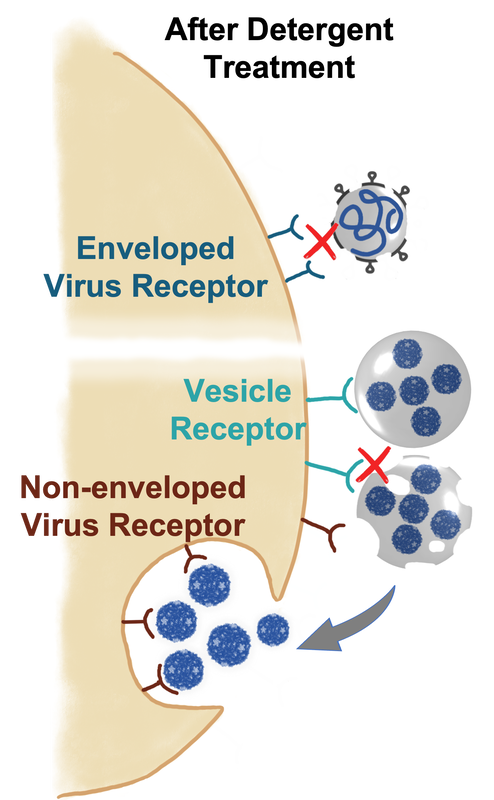
Vesicle-cloaked virus clusters blur the distinction between enveloped and non-enveloped viruses but behave like none of them. For example, enveloped viruses are susceptible to detergent treatment because they loses fusion capability after the treatment. However, some persistent viral vesicles could survive after detergent treatment. Even though the vesicle membrane is destructed, the non-enveloped viruses still remain infectious after released from the vesicles. |
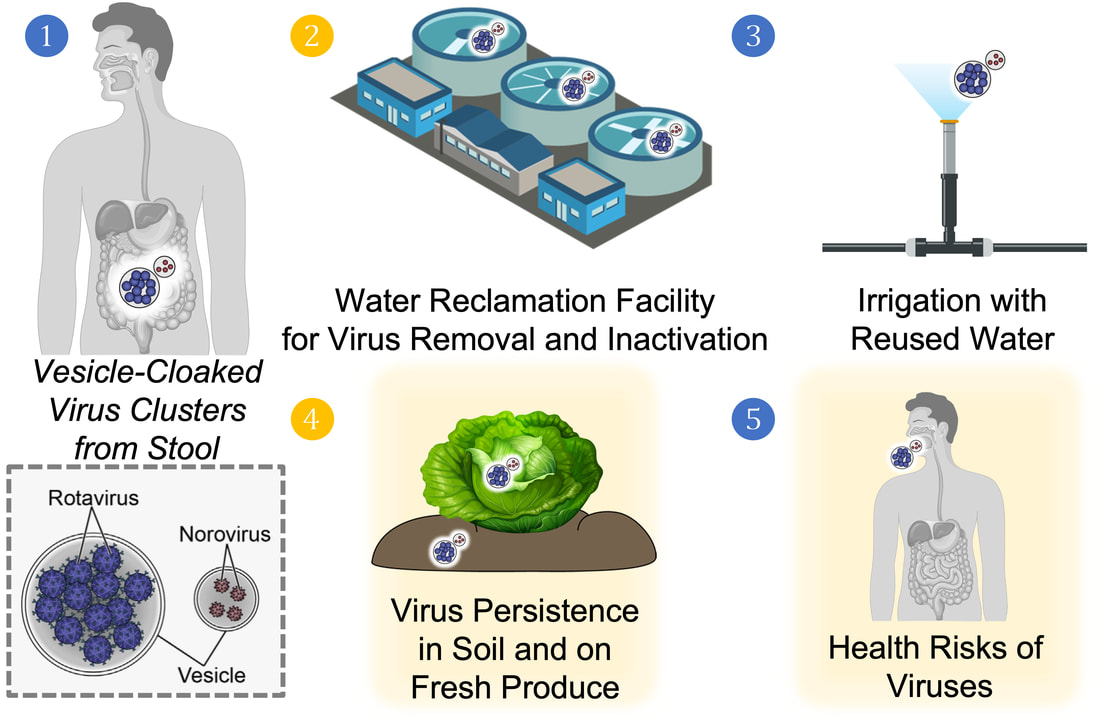
Persistence and Inactivation of Vesicle-Cloaked Enteric Virus Clusters in Agricultural Reuse Water (Funded by USDA-NIFA Water Quantity and Quality Program, 2022-2024)
|
With an increasing demand of freshwater for agricultural irrigation, the reuse of wastewater, an alternative water source, has emerged as a promising practice to provide sufficient and safe water. However, the persistence and inactivation of viral vesicles in wastewater treatment for reuse and on the farmland and crop surfaces after irrigation is unknown.
We aim to understand the removal and inactivation of rotavirus and norovirus vesicles in water reuse, particularly membrane filtration and advanced oxidation processes, and evaluate the persistence of rotavirus and norovirus vesicles in soil and on crop surface irrigated with reused water. |
The project scopes are different and there is no overlap between the projects. The NSF project focuses on the environmental implication of viral vesicles in water system, the NIH project explores the engineering control of the viral vesicles in built environment, and the USDA-NIFA project evaluates the environmental implication of the viral vesicles in agricultural and food systems. Taken all together, these projects will provide complementary and comprehensive insights on the environmental and biological behavior of vesicle-cloaked enteric virus clusters in different environment media, i.e., water, food, contact surfaces, as well as control strategies for inactivating these emerging pathogens to protect public health.